 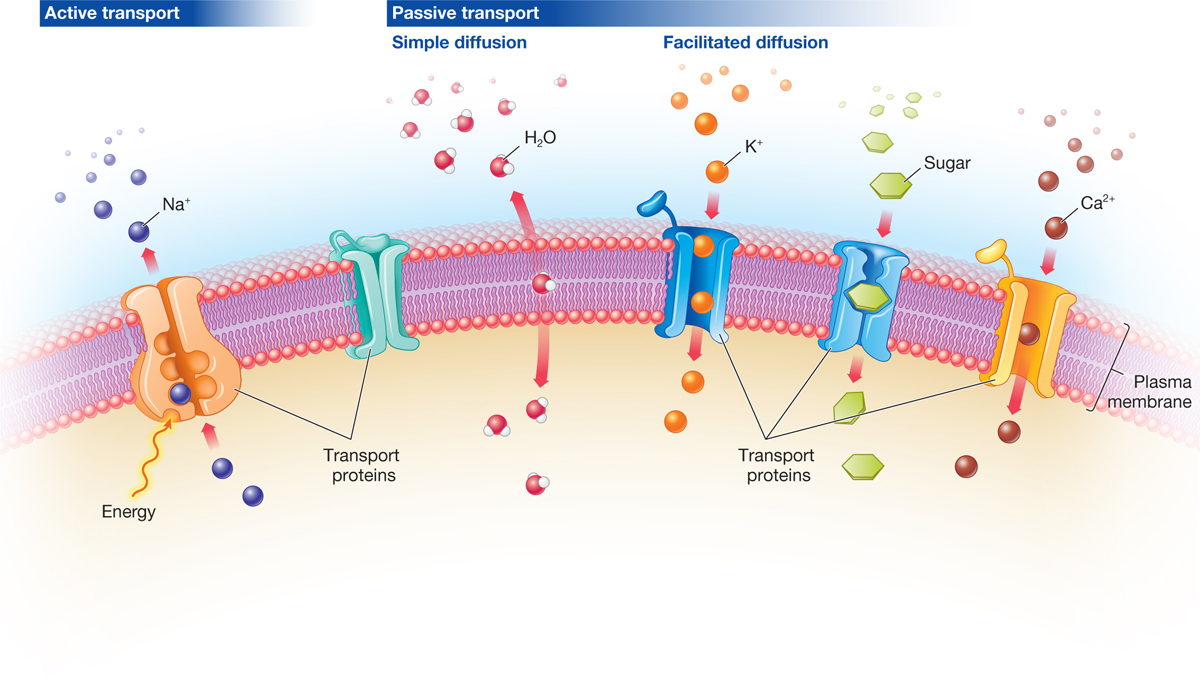 
The reasons should be self-evident: larger molecules simply cannot fit between the lipid molecules to make their way through. However, small nonpolar molecules are able to pass through the membrane with relative ease. ethanol), but more often pass at low rates if at all (e.g. Small polar molecules can sometimes pass easily (e.g. 4.2: Membrane Permeability A pure phospholipid bilayer, whatever the lipid composition, is a semi-permeable membrane that is generally repellent to large molecules and to ions.Cellular membranes were partially defined as being composed primarily of phospholipids: molecules consisting of a phosphorylated polar head group attached to a glycerol backbone that has two long hydrocarbon tails. 4.1: Membrane Structure and Composition Since most cells live in an aqueous environment and the contents of the cell are also mostly aqueous, it stands to reason that a membrane that separates one side from the other must be hydrophobic to form an effective barrier against accidental leakage of materials or water. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
